19A178
Initial Analysis of Prescribing Trends on PCRS Hub* (*At ISR, updated quarterly data will be presented from June 1st to September 1st 2019)
Author(s)
C Kirby (1), G Killeen (2), RH Mullan (1), D Kane (1,2)
Department(s)/Institutions
Tallaght University Hospital (1) & HSE National Clinical Programme for Rheumatology (2)
Introduction
The ‘High Tech Prescription Hub’ (HTPH) was established on June 1st for prescribing biologic DMARDS. The HTPH identifies ‘Best Value Biologics’ (BVB, lowest cost biosimilar) to the prescriber for adalimumab and etanercept to promote initiation and switching.
Aims/Background
Data from the HTPH on biologic prescribing practices in Ireland is presented.
Method
All prescriptions issued by rheumatology, gastroenterology and dermatology on the HTPH from June 1st to July 15th were analysed by therapeutic agent, specialities and hospital groups. *
Results
Of 40 individual hospital sites in Ireland, registration rates for HTPH were as follows: rheumatologists 33/40, gastroenterologists 16/40, dermatologists 10/40. Of the 7 sites with no rheumatologist registered, the local rheumatologist was already registered at another hospital site, giving 100% registration for rheumatology.
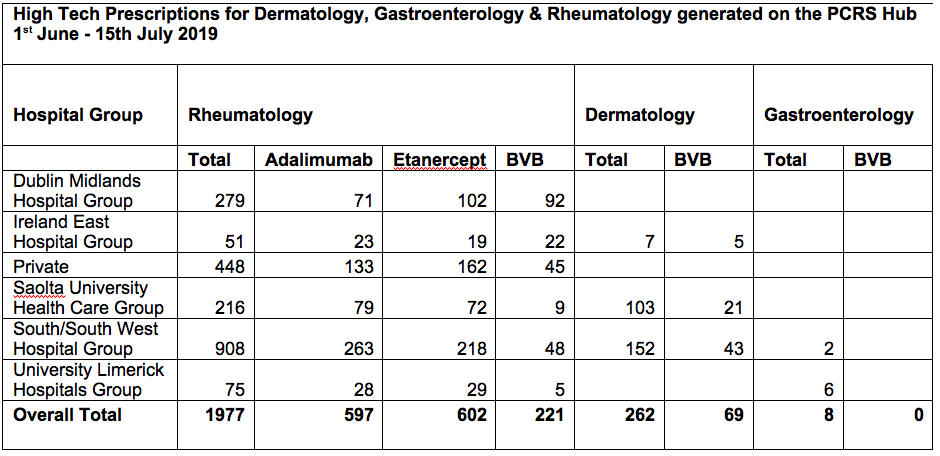
2247 HTPH biologic prescriptions were issued between June 1st and July 15th - 1977 (88%) by rheumatology, 262 (11%) by dermatology and 8 by gastroenterology (<1%). For Rheumatology prescriptions, South/Southwest Hospital Group (SSWHG) accounted for 908 (46%) prescriptions followed by Private Hospitals (PH) with 448 (22.7%), Dublin Midlands Hospital Group (DMHG) with 279 (14%), Saolta University Healthcare Group (SUHG) with 216 (11%), University Limerick Hospitals Group (ULHG) with 75 (4%) and Ireland East Hospital Group (IEHG) with 51 (2.6%).
Adalimumab was prescribed for 597 patients in rheumatology (30% of all rheumatology prescriptions). Humira was prescribed in 473 (79%) cases, Imraldi in 76 (12.7%), Amgevita in 43 (7.2%) and Hulio in 5 (0.83%) cases. Etanercept was prescribed in 602 rheumatology patients (31% of all rheumatology prescriptions) with 150 (25%) being prescribed Benepali.
In all, 290 patients were switched to, or initiated on, a BVB: rheumatology = 221 (76.2%), dermatology = 69 (23.7%) and gastroenterology = 0. BVB prescriptions in rheumatology are as follows: DMHG 92 (41.6%), SSWHG 48 (22%), PH 45 (20.4%), IEHG 22 (10%), SUHG 9 (4%), ULHG 5 (2.3%).
Conclusions
Adalimumab and etanercept comprised almost 2/3 of all biologic prescriptions in rheumatology. Uptake of biosimilars since June 1st 2019 is 22% with biosimilar prescription rates varying between hospital groups and specialties. Rheumatology accounts for the vast majority of biosimilar prescriptions in the initial period of HTPH operation.